Editor's Choice
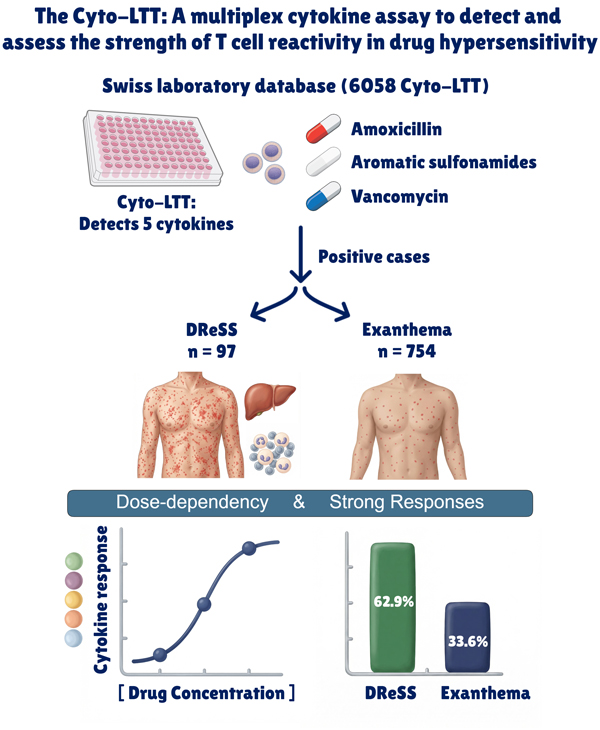
Editor’s comment: The conventional in vitro lymphocyte transformation assay (LTT) for diagnosing drug hypersensitivity reactions is limited by sensitivity. Peyer et al. (Switzerland) report a cytokine-based lymphocyte transformation test (Cyto-LTT) that quantifies drug-specific cytokine responses in T cells. The results reveal dose-dependent T cell activation in delayed drug hypersensitivity, challenging that drug allergic reaction are dose-independent. Their findings suggest that delayed drug hypersensitivity reflects graded immune activation with clinical relevance. Please also refer to Review Article by Werner J. Pichler entitled "Undressing DReSS as p-i mediated disease"
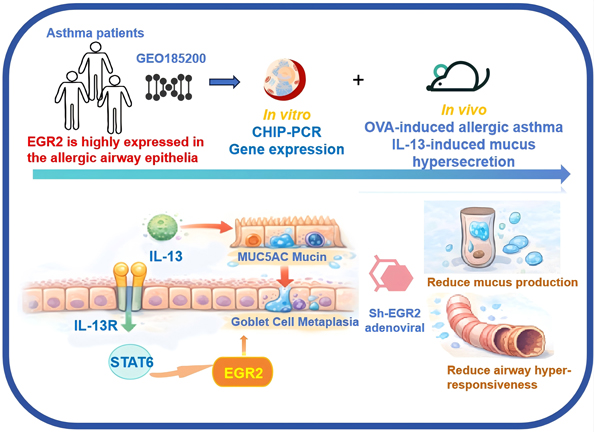
Editor’s comment: Goblet cell metaplasia is a key pathological feature of allergic asthma, but its regulatory mechanisms remain incompletely defined. Xu et al. (China) demonstrates that transcription factor EGR2 is selectively upregulated in allergic airway epithelium and functions downstream of IL-13/STAT6 signaling. Genetic and pharmacological modulation of EGR2 alters mucus production and airway hyperresponsiveness, identifying EGR2 as a critical transcriptional regulator of asthmatic epithelial remodeling.
Review Series: An evolving understanding of drug allergy: Novel basic mechanisms and their clinical impact
Recent progress has greatly changed our understanding of basic mechanisms and clinical aspects in drug allergy. In this issue of Allergology International, three groups of scientists and clinicians involved in drug allergy have contributed to a review series entitled An evolving understanding of drug allergy – novel basic mechanisms and their clinical impact. A sophisticated approach for basic and clinical aspects of drug allergy will definitely enhance the clinical unraveling of immunological mechanisms, clinical assessment, and prompt management. These findings collectively enhance the clinical impact on drug allergies, saving the lives of ADR patients.
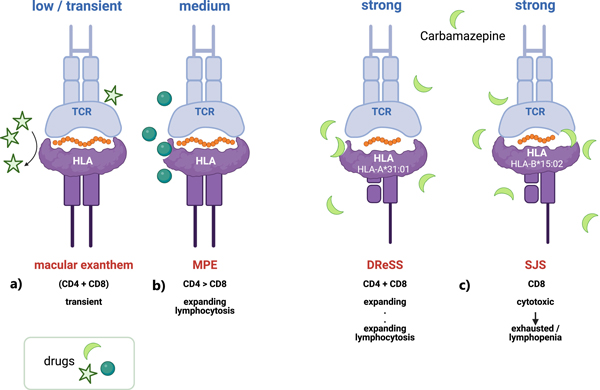
Pichler et al. wrote one review article and one original paper for this issue. Long ago, the authors proposed the p-i mechanism (pharmacological interaction with immune receptors), now widely accepted, to explain T cell activation. Drug reaction with eosinophilia and systemic symptoms (DReSS), also called drug-induced hypersensitivity syndrome (DIHS), is a severe T cell-mediated hypersensitivity reaction, presumably involving a distinct p-i mechanism. In the review article, they propose four phases based on the status of p-i stimulated T cells: the clinically silent phase, the acute phase, the viraemic and autoimmunity phase, and the chronic phase.
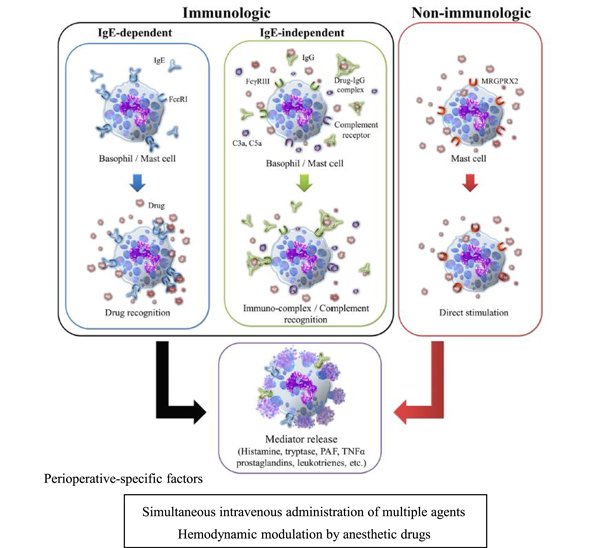
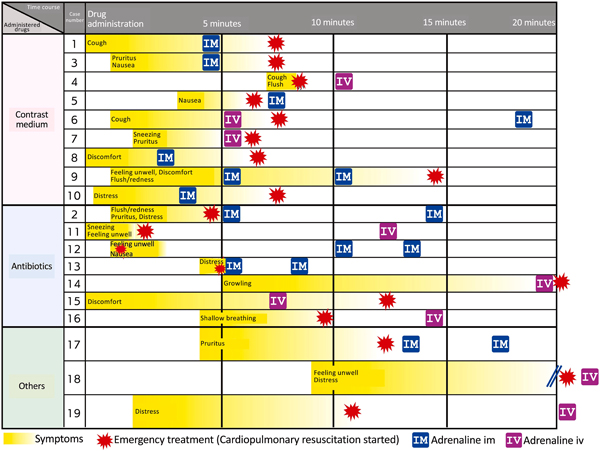
Takazawa has realized a powerful approach into perioperative anaphylaxis, occurring in approximately 0.01 to 0.02% of all subjects receiving general anesthesia.6 The Japanese Epidemiological Study for Perioperative Anaphylaxis (JESPA) prospectively analyzed perioperative anaphylaxis cases and control cases; perioperative anaphylaxis can correctly be diagnosed based on clinical findings and tryptase measurement.
In Yamaguchi et al. article, they mention governmental activities related to drug-induced deaths related to anaphylactic shock. The review describes how prompt adrenaline use is realized in clinical situations in Japan, probably reflecting nationwide notification of the importance of prompt diagnosis and treatment using intramuscular adrenaline. A successful management approach may be useful for suppressing deadly episodes of anaphylactic shock.