Volume 69, Issue 2 (April 2020)
Editor's Choices
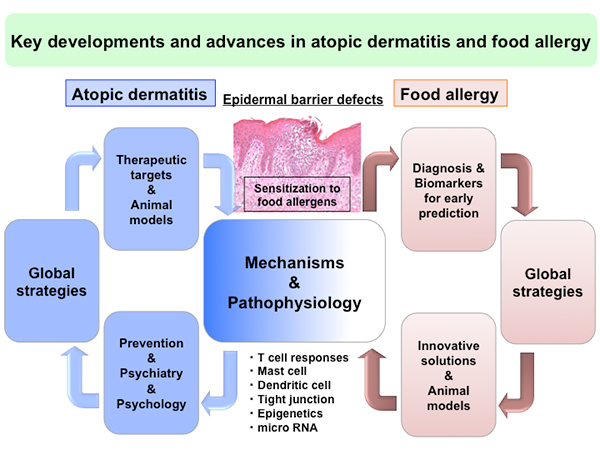
Editor’s comment: Dr. Sugita and Prof. Akdis summarized recent advances in atopic dermatitis (AD) and food allergy (FA), particularly with regard to molecular mechanisms and disease endotypes of AD and FA, also including recent developments in global strategies for patient management. I believe that this review article will greatly contribute to our readers' understanding of the pathogenesis of AD and FA and of strategies for their treatment.
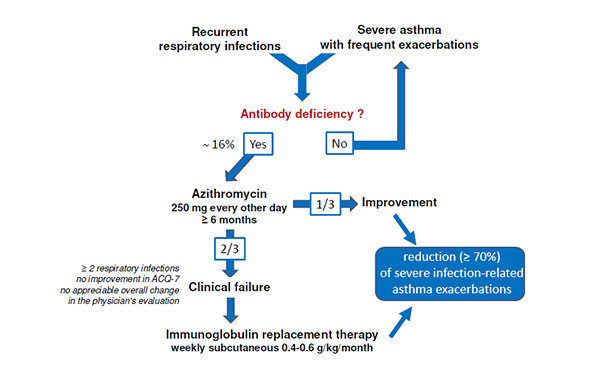
Editor’s comment: Antibody deficiencies such as selective IgA deficiency and IgG subclass deficiencies are associated with a risk of asthma exacerbation. Although antibody deficiencies are well known causes of recurrent respiratory infections, little is known about their impact on adults with asthma. In France, Tiotiu et al. performed a five-year prospective observational study of adults with severe asthma and antibody deficiency to assess treatment outcomes with either azithromycin or subcutaneous immunoglobulins. They showed that both of these treatments dramatically improved asthma outcomes, suggesting that adults with severe asthma and recurrent respiratory infections should be screened for antibody deficiency and treated with azithromycin or subcutaneous immunoglobulins as appropriate.
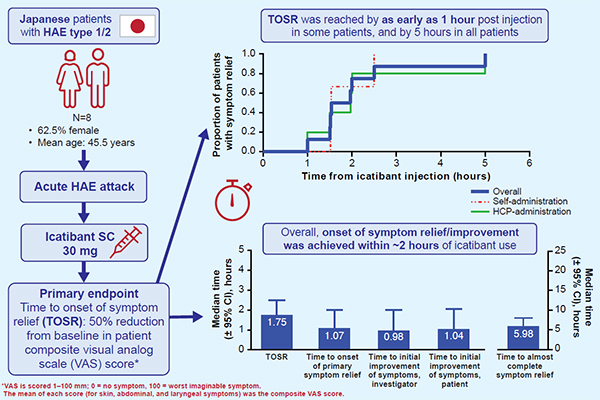
Editor’s comment: Hereditary angioedema (HAE) is an autosomal-dominant genetic disease characterized by recurrent swelling episodes affecting the skin, gastrointestinal mucosa, and upper respiratory tract. HAE is mainly caused by a loss of functional C1 inhibitor (C1-INH), leading to activation of the kallikrein-kinin system and a subsequent increase in circulating bradykinin levels, which mediate the clinical symptoms of HAE. Hide et al. conducted a phase 3, single-arm, open-label study to evaluate icatibant, a selective bradykinin B2 receptor antagonist, for the treatment of acute attacks in Japanese patients with HAE. Although the number of patients was small, the efficacy and tolerability of icatibant for acute attacks were demonstrated, whether self-administered or administered by a healthcare professional.
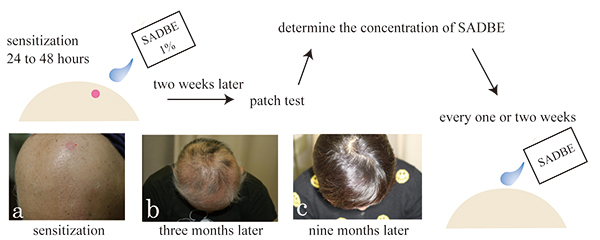
Editor’s comment: Although the Japanese guidelines for the treatment of alopecia areata include topical immunotherapies, evidence of its efficacy remains insufficient. Sasaki et al. performed a retrospective cohort study on 49 Japanese alopecia patients who had received topical immunotherapy with squaric acid dibutylester (SADBE) to clarify the effect of this topical immunotherapy on the progression and severity of their alopecia areata. They showed that the improvement rate in all alopecia patients treated with SADBE topical immunotherapy was over 50%, supporting the recommendation of this treatment for alopecia areata in the Japanese guidelines.